
Best Supplements for Pineal Gland Health: Ranked 1–10 by Evidence Score
10 pineal gland supplements ranked by evidence score — not marketing claims. Magnesium scored 8.4/10. Most popular products ranked lower...

Most people trying to decalcify their pineal gland focus on what to remove. Detox protocols. Filtered water. Eliminating fluoride from the diet. All of that matters, but it misses something.
There’s a mineral that may work from the other direction.
Boron is a trace mineral that may help decalcify the pineal gland by acting as a fluoride antagonist — blocking the enzyme pathways that allow fluoride to bind to soft tissue and form calcified deposits. It doesn’t flush fluoride. It competes with it. That’s a different mechanism, and in some ways a more targeted one.
If you’ve already read about fluoride and the pineal gland, you know that fluoride doesn’t just affect teeth. It accumulates in the pineal gland at concentrations higher than in bone. Boron research suggests there may be a way to interfere with that process biochemically.
Here’s what the evidence actually shows — and where it runs out.

Boron is a trace mineral found in plants, fruits, nuts, and legumes. It’s not classified as an essential nutrient in the same way iron or calcium are, but research has quietly been building a case for its biological relevance since the 1980s.
Its established roles involve mineral metabolism: boron regulates how the body handles calcium, magnesium, and phosphorus. When boron is deficient, calcium excretion increases and bone density drops. That relationship with mineral balance is exactly why researchers started asking questions about its interaction with fluoride, another mineral that competes for the same biochemical territory.
The pineal gland, a small calcite-heavy structure already prone to mineralization, sits at the center of that conversation.
Boron may protect the pineal gland from fluoride-induced calcification by interfering with the biochemical process that causes it. The mechanism isn’t simple, and it isn’t fully confirmed in humans, but the biochemistry is coherent.
Boron competes with fluoride for binding sites in soft tissue — a mechanism documented in a 1999 paper by Naghii published in Nutrition and Health showing that boron modulates fluoride retention and distribution throughout the body.
The core mechanism: certain enzymes facilitate the binding of fluoride to calcium in soft tissue. Boron appears to inhibit those pathways. When boron occupies those binding sites first, fluoride has fewer places to attach, which may reduce the rate at which fluoride accumulates in tissues like the pineal gland.
Research suggests. May interfere with. Those qualifiers are intentional. Naghii’s work examined metabolic modulation broadly. It wasn’t a pineal-specific study. But the mechanism it describes maps directly onto what we know about how fluoride calcifies the pineal gland in the first place.
Here’s what’s actually happening at the molecular level.
Fluoride doesn’t calcify tissue on its own. It binds to calcium ions to form a compound called fluorapatite, a crystalline structure that deposits in soft tissues over time. The pineal gland, with its uniquely high blood flow and calcium-rich environment, is especially susceptible.
Boron interferes with that binding. By competing with fluoride for the calcium-binding sites in soft tissue, it may reduce the rate of fluorapatite formation. Less fluorapatite. Less calcification.
That’s the hypothesis. It’s biochemically plausible. The gap, and the curator was clear about this, is that no study has tested it directly in pineal tissue. The mechanism is inferred from bone and soft tissue research. Not invented. Inferred.
That distinction matters.
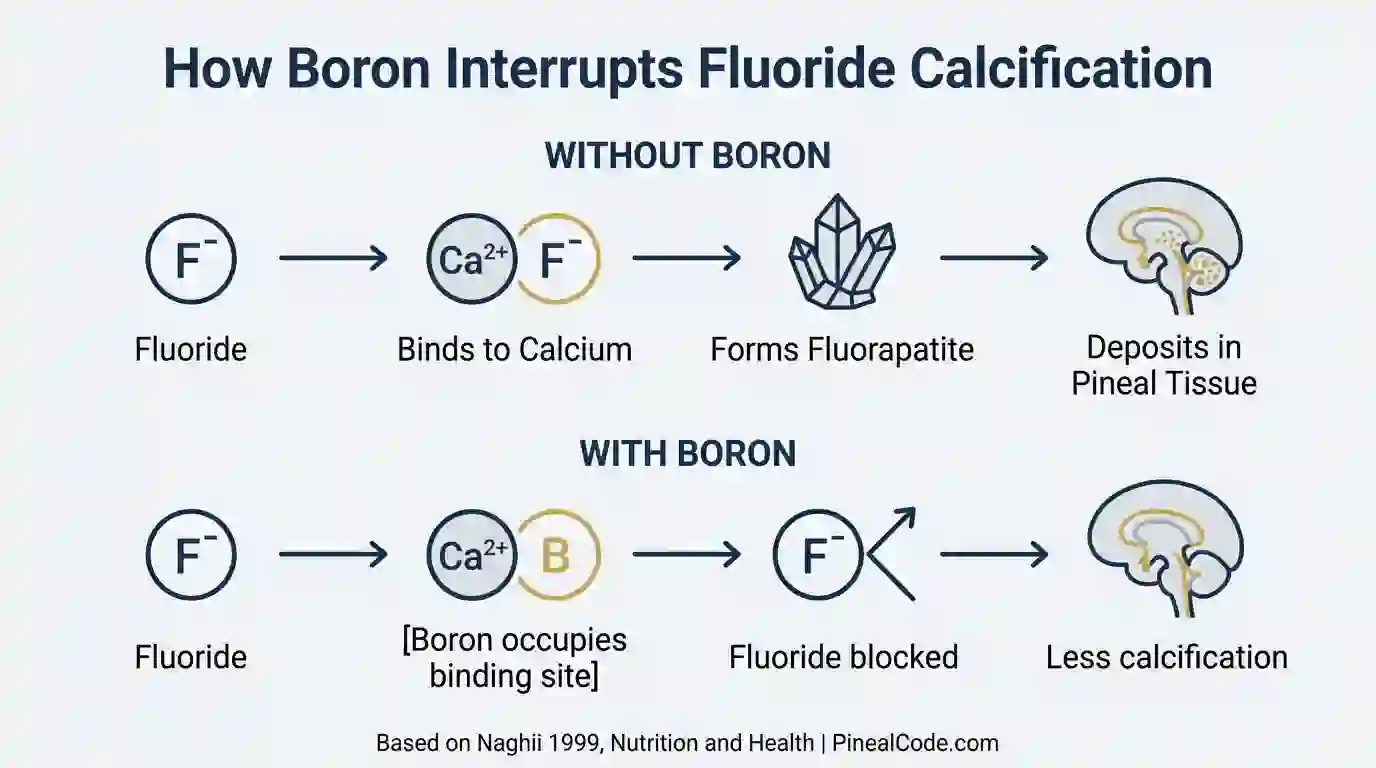
Direct evidence is limited. That’s the honest answer, and honest is more useful here than confident.
No randomized controlled trial has measured boron’s effect specifically on pineal gland calcification in humans. The research connecting boron to pineal decalcification is built from two separate bodies of evidence that haven’t been tested together: studies showing fluoride accumulates in the pineal gland (Luke et al., 2001, Caries Research) and studies showing boron modulates fluoride metabolism in the body (Naghii, 1999; Nielsen et al., 1987).
The indirect evidence is actually interesting. A 2013 EFSA safety assessment on boron confirmed its role in mineral regulation, noting effects on calcium and fluoride metabolism across multiple tissue types. Nielsen’s FASEB Journal work from 1987 showed that 3 mg/day of boron significantly reduced urinary calcium excretion. That’s evidence that boron is biologically active at supplemental doses.
But “biologically active” and “decalcifies the pineal gland” are not the same sentence.
I’ll be honest: when I first started tracking boron research, I thought the fluoride-antagonist mechanism was elegant enough to be practically conclusive. Then I looked harder at what “indirect evidence” actually means here. It means we’re inferring from adjacent data. That’s different from confirmation.
Boron is probably the most mechanistically specific agent available against fluoride-driven pineal calcification. That’s a real distinction compared to other supplements in this space, which work through less targeted pathways, as covered in how to reverse pineal gland calcification. Specific isn’t the same as proven.
Work with what we have. Know what we don’t.
Diet can get you part of the way there. Not all the way, but the baseline matters.
| Food | Boron per 100g (approx.) |
|---|---|
| Raisins | 1.0–3.0 mg |
| Almonds | 1.5–2.5 mg |
| Avocado | 1.0–2.0 mg |
| Prunes | 1.0–2.0 mg |
| Walnuts | 1.0–2.0 mg |
| Honey | 0.5–1.0 mg |
| Chickpeas | 0.7–1.5 mg |
These numbers come from USDA Agricultural Research Service data and Meacham et al. (Plant Foods for Human Nutrition, 1994). Raisins are consistently one of the densest dietary sources, which is either a convenient fact or nature’s strangest supplement recommendation.
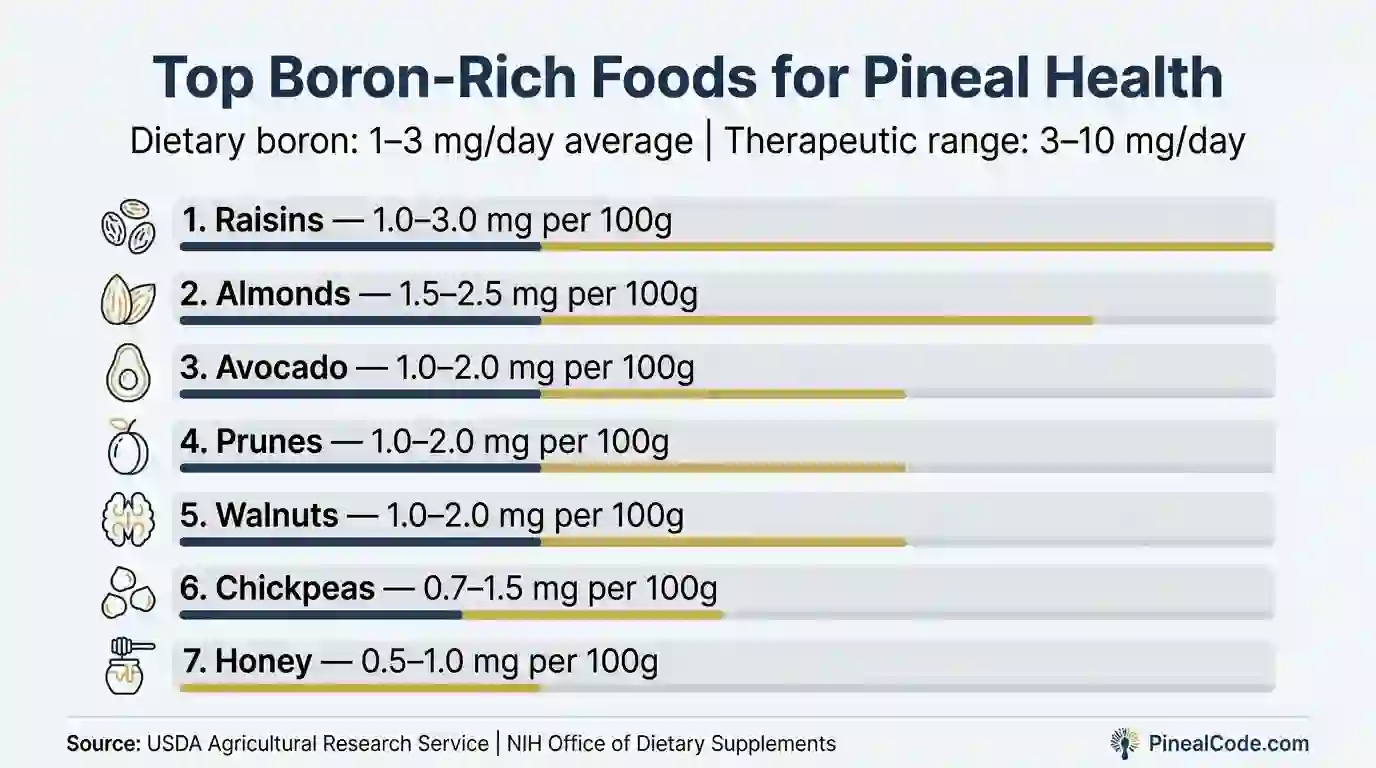
A reasonably balanced diet in the U.S. and Europe delivers about 1–3 mg of boron per day. That keeps you out of deficiency. It won’t get you to the 3–10 mg range associated with fluoride antagonism in the research literature.
Rex Newnham, an Australian botanist who spent the 1960s self-experimenting with boric acid for his arthritis before anyone in mainstream medicine took him seriously, would recognize this pattern. Useful compounds rarely arrive with proper credentials.
For therapeutic doses, you need supplementation. The most studied forms are boron citrate (generally preferred for bioavailability), sodium borate, and calcium fructoborate. Absorption across these forms runs above 85% in available studies, which means the bottleneck isn’t getting boron into the bloodstream. It’s whether enough crosses into the tissues that matter.
That’s a real open question. Worth saying out loud.
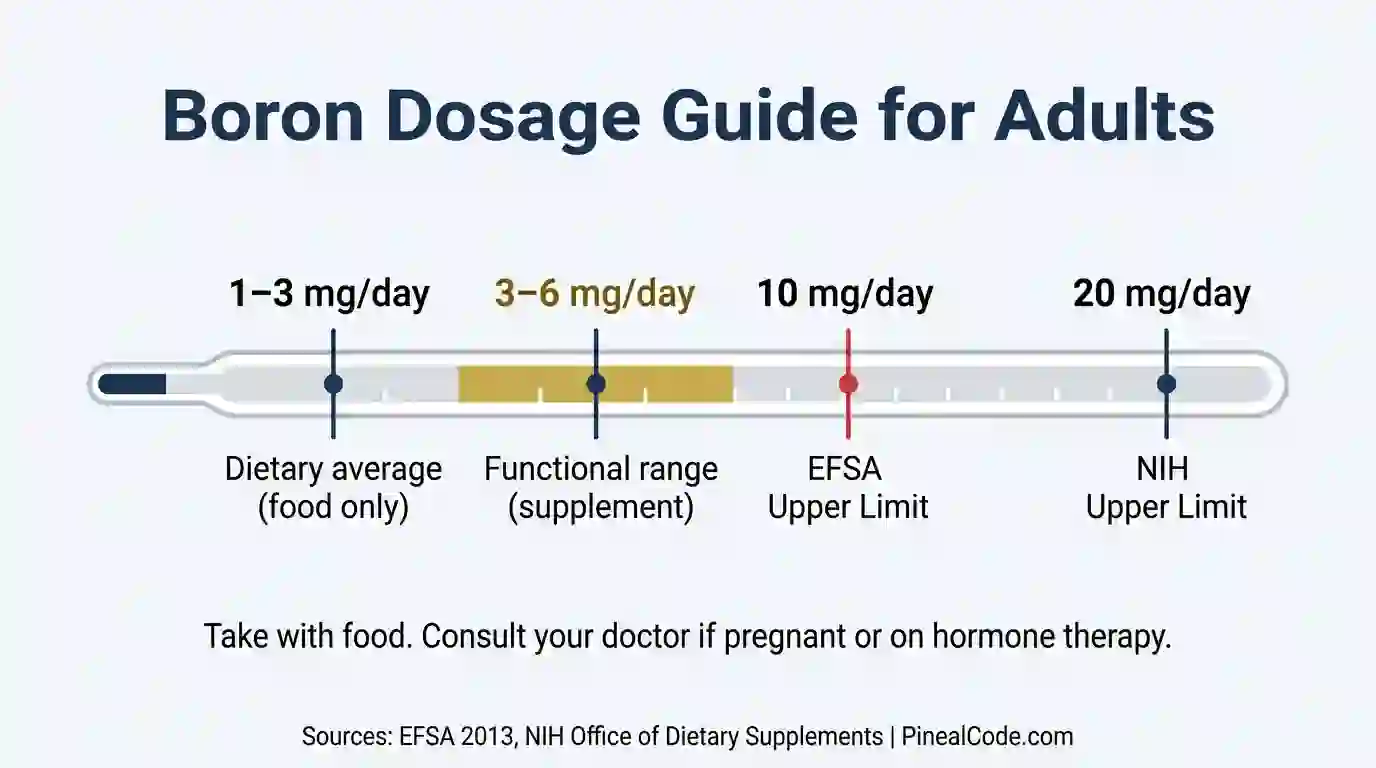
The functional range in research is 3–10 mg per day. Most commercially available boron supplements sit at 3–6 mg per dose, which aligns with the doses used in the Nielsen mineral metabolism studies.
Two safety reference points:
The gap between those numbers reflects different methodological approaches to safety assessment, not a discrepancy in the underlying data. Conservative approach: stay at or below the EFSA threshold of 10 mg/day. Take it with a meal. Boron on an empty stomach occasionally causes mild gastrointestinal discomfort.
This is not medical advice. Talk to your healthcare provider before starting any supplement protocol, especially at higher doses.
Three populations need a harder look before supplementing:
Boron doesn’t work as a standalone intervention. Nothing in this space really does.
The protocols that make biochemical sense combine boron with nutrients that address different parts of the calcification process. A stack that shows up consistently in both Nielsen-era mineral research and the r/nootropics protocols that have been circulating for the past decade:
Timing: with your main meal. Duration: minimum 90 days. Biochemical changes in mineral metabolism are slow. They don’t show up in two weeks. Anyone claiming otherwise is selling something.

Take this with your largest meal. Run it for at least three months before drawing conclusions. And pair it with everything covered in the best supplements to decalcify your pineal gland and pineal gland decalcification at night.
If you’re looking for a formulated supplement that combines boron with other pineal-supportive ingredients, Pineal Guardian and PinealPure are two options worth reviewing.

Boron is the most mechanistically specific tool we currently have against fluoride-driven pineal calcification. That’s not nothing. Most supplements in this category work through general detox pathways. Boron targets the biochemical process that turns fluoride into calcified tissue in the first place.
But specific isn’t proven. No RCT has tested this in human pineal tissue. The mechanism is built from sound indirect evidence, and evidence built from inference is still evidence, just not certainty.
One thing worth pushing back on before you close this tab: most content around pineal decalcification implies that clearing calcification will unlock something. More vivid dreams. Sharpened awareness. A generally more interesting inner life. That framing runs well ahead of the evidence. What the research actually supports is that calcification correlates with reduced melatonin output in aging adults. That’s a real physiological outcome worth addressing on its own terms, without the spiritual overlay. The overlay is usually selling something.
If you’re building a serious decalcification protocol, boron belongs in it. At 3–6 mg/day, taken with food, within EFSA safety parameters. Not a miracle solution, but one piece of a 90-day minimum commitment alongside magnesium, D3, and K2.
Start there. Adjust based on what you notice. Everything about building a complete protocol is in how to decalcify your pineal gland naturally.
Marcus Hale is an independent researcher and former clinical neuroscientist. The content on PinealCode.com is for informational purposes only and does not constitute medical advice.

10 pineal gland supplements ranked by evidence score — not marketing claims. Magnesium scored 8.4/10. Most popular products ranked lower...
One peer-reviewed study found 9,000 ppm of fluoride in human pineal glands — 18,000x more than in muscle. Here's what that data means, what...
61.65% of adults have pineal calcification — and the internet promises a 30-day fix. Actual research timeline: 3–6 months. Week-by-week...

Marcus Hale
Independent Researcher · Former Clinical Neuroscientist
I spent 12 years in clinical neurology before the questions got more interesting than the answers. PinealCode is where I document what I find at the intersection of brain science and consciousness.