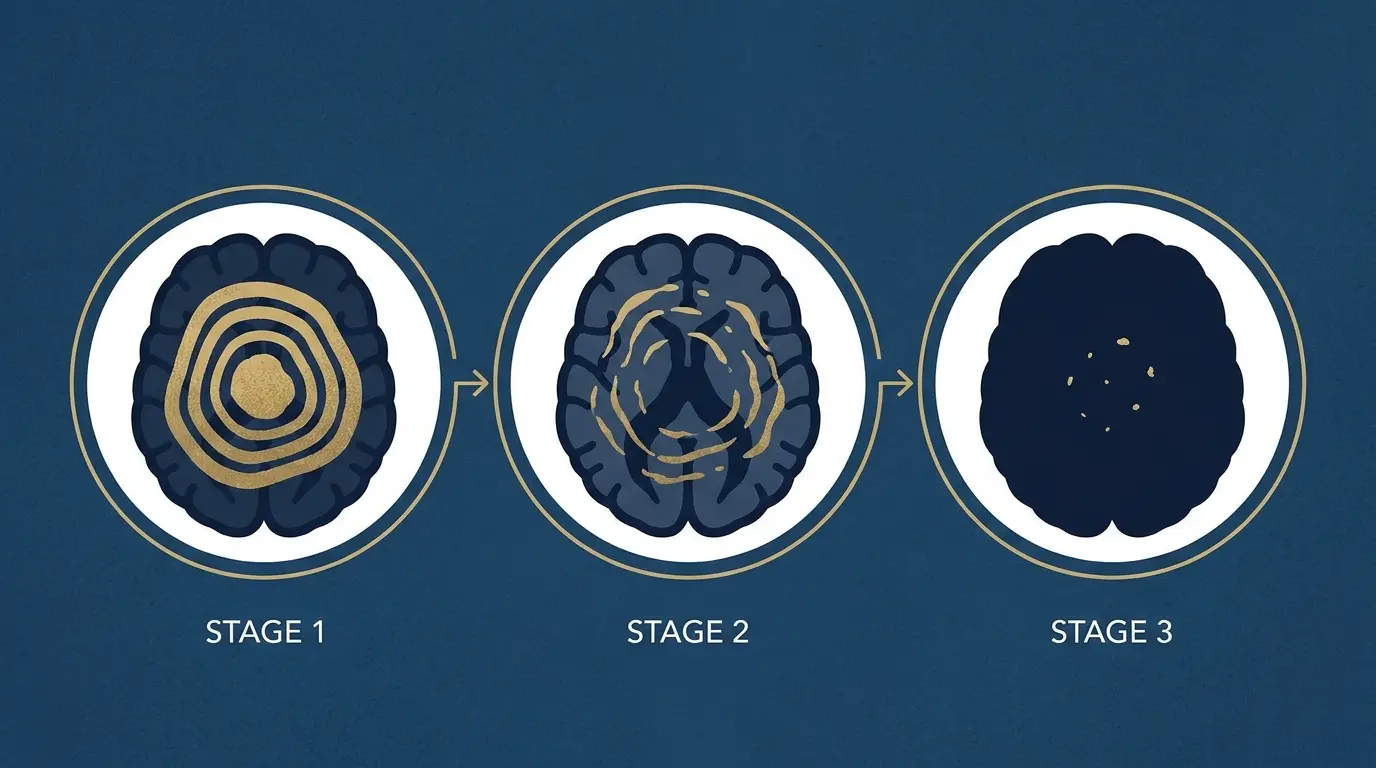
How Long to Decalcify Your Pineal Gland? Not 30 Days
61.65% of adults have pineal calcification — and the internet promises a 30-day fix. Actual research timeline: 3–6 months. Week-by-week...

The wellness internet has decided that bentonite clay decalcifies your pineal gland. Thousands of people are mixing gray powder into water every morning because of it.
The actual chemistry tells a different story. Not a worse story. More precise.
Bentonite clay is a real thing with real mechanisms. It binds heavy metals in the digestive tract. That’s documented, peer-reviewed, not controversial. The question is whether “binds heavy metals in the gut” equals “decalcifies a gland in the center of your brain.” Those are not the same claim. They’re not even in the same category.
Here’s what the evidence actually shows. And where it runs out completely.
No — not directly, and the chemistry explains why. Bentonite is an aluminosilicate clay that works through cation exchange: it carries a negative surface charge and attracts positively charged metal ions like lead, mercury, aluminum, and cadmium. Pineal gland calcification is primarily calcium hydroxyapatite: a stable crystalline calcium phosphate compound. That’s a different chemical target entirely. Bentonite’s adsorption mechanism doesn’t dissolve hydroxyapatite deposits. Structurally, it can’t.
But here’s where it gets more interesting than a flat “no.”
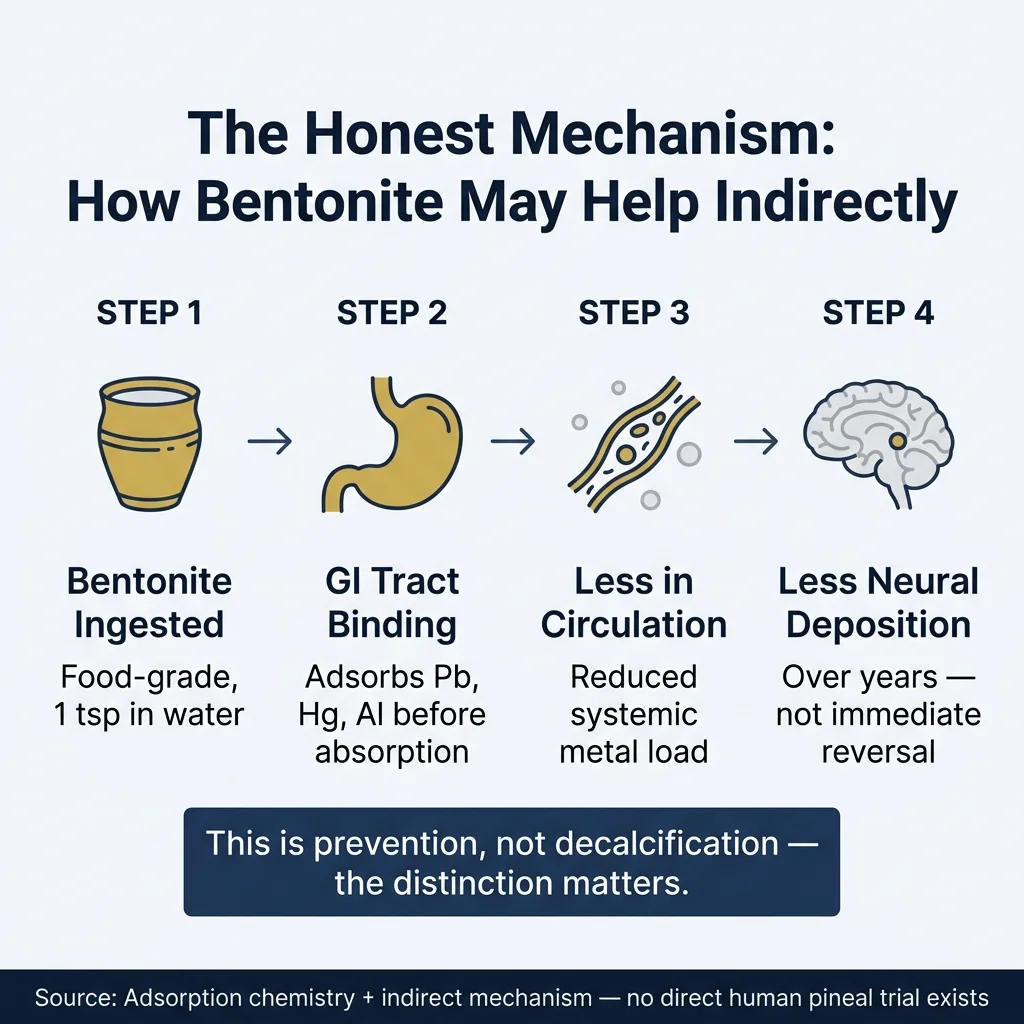
Heavy metals, particularly lead and aluminum, are found in elevated concentrations in calcified pineal tissue. The 2002 work from Baconnier et al. identified distinct crystal formations in human pineal epithelium, and the broader literature on pineal calcification implicates metal accumulation as a contributing factor alongside fluoride and aging. If bentonite clay reduces the systemic load of those metals before they reach neural tissue, intercepting them in the GI tract before absorption, that’s not decalcification. That’s upstream prevention.
Prevention and reversal are different things. Be precise about which one you’re pursuing.

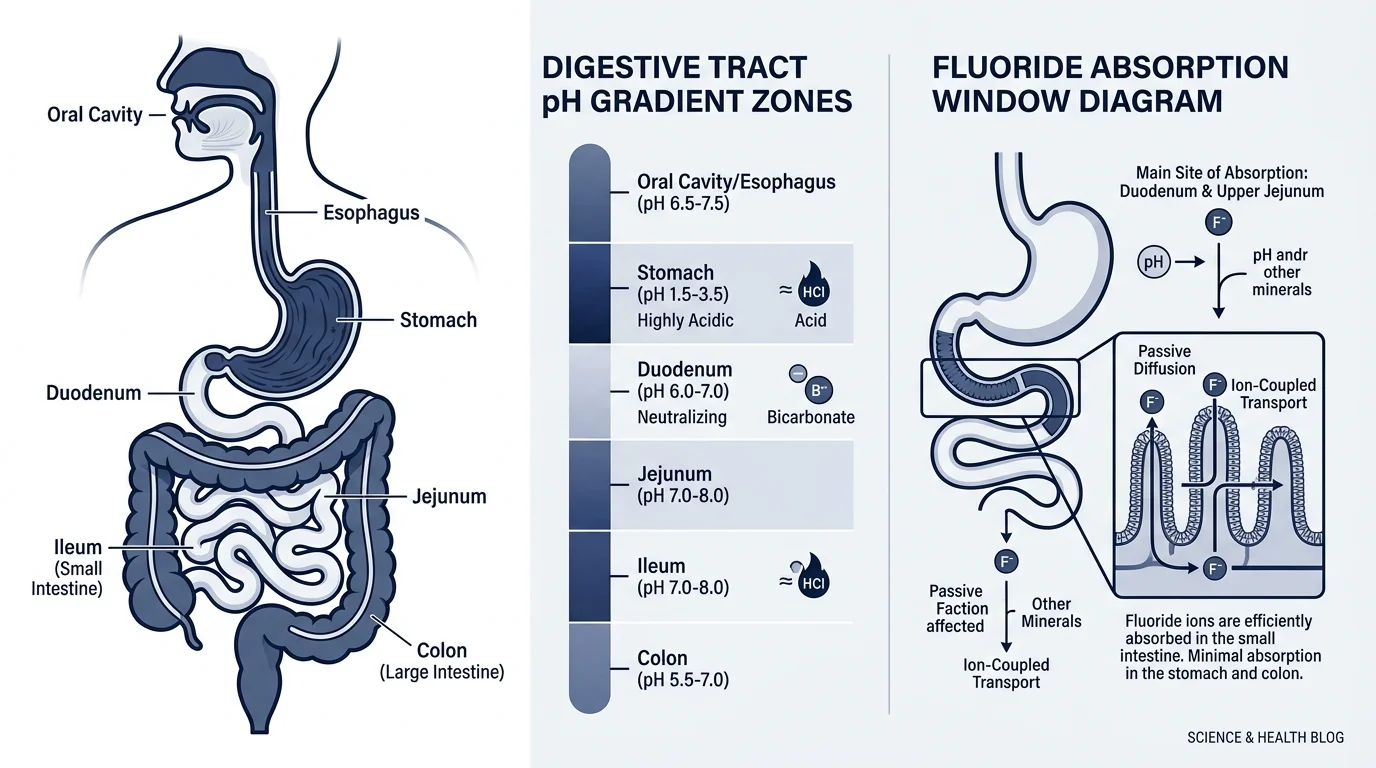
Partially, and only under specific conditions. In water purification studies, modified bentonite achieves fluoride adsorption, but optimal efficiency occurs at pH 3, which corresponds to stomach-level acidity. By the time bentonite reaches the small intestine (pH 6–7.5), fluoride binding capacity drops significantly.
More important: fluoride already incorporated into hydroxyapatite in pineal tissue is chemically bound into a crystal lattice. Bentonite in your digestive tract cannot reach it. That fluoride is gone. Structurally integrated. The window where bentonite could theoretically intercept fluoride is during absorption, before it enters circulation. Whether that actually reduces pineal fluoride burden in humans has never been measured.
Bentonite clay reduces fluoride absorption from water at stomach pH, but cannot remove fluoride already crystallized into calcium hydroxyapatite deposits in the pineal gland — a distinction no human clinical trial has yet quantified.
That gap in the literature isn’t a conspiracy. It’s just that nobody has funded this study. It probably won’t make anyone money.
Descartes called the pineal gland the seat of the soul. Three hundred years later we’re still not fully sure what it does beyond melatonin production. We are fairly sure we’d prefer it uncalcified.
Lead, mercury, and aluminum accumulate preferentially in neural tissue. The brain is 60% fat, and certain heavy metals have a strong affinity for lipid-rich environments. The pineal gland, small, heavily vascularized, and sitting outside the blood-brain barrier in several physiological models, is particularly exposed.
Pineal gland calcification occurs in approximately 61.65% of adults, according to a 2023 systematic review and meta-analysis, with rates rising predictably with age. That’s not a fringe statistic. It’s one of the most commonly calcified structures in the human brain.
Here’s what bentonite can plausibly do: a 2025 study in Scientific Reports showed activated calcium bentonite adsorbs up to 13 mg/g of lead and 14 mg/g of copper from aqueous solution. These are water treatment numbers, not human gut numbers. But the mechanism is real. If even a fraction of that binding capacity operates in the GI tract, you’re reducing the amount of lead and aluminum entering circulation in the first place.
Less circulating lead. Less available for neural deposition. Less potential contribution to calcification over decades.
That’s the honest version of the claim.
There’s a paradox worth sitting with, though. Bentonite’s chemical formula is Al₂O₃·4SiO₂·H₂O. It naturally contains aluminum as a structural component. Whether food-grade bentonite releases bioavailable aluminum during GI transit, and whether that offsets any heavy metal binding benefit, has not been adequately studied in humans. Nobody can give you a clean answer on this yet.
Here’s what I can say with reasonable confidence: the benefit mechanism is real. Reduced GI absorption of heavy metals is biologically sound and backed by adsorption chemistry, even without direct human data.
The risk side is trickier. Aluminum leaching from the clay itself is plausible and equally unquantified. I genuinely don’t know which effect dominates in practice. Neither does anyone else, because the study hasn’t been done.
What we do know is that food-grade calcium bentonite has established safety for short-term oral use as an enterosorbent, with the most rigorous safety data coming from aflatoxin-binding research out of Texas A&M. That’s not the same as a long-term heavy metal protocol. Different dose, different duration, different intended mechanism.
If you’re going to use it, the grade distinction matters more than the brand.
One teaspoon of food-grade calcium bentonite stirred into 8–12 oz of water. Drink it, don’t chew it, which sounds obvious until you’ve seen what people do online. Use a non-metal spoon; the clay interacts with metal utensils. Let it sit a few minutes to hydrate before drinking.
Take it at least 1–2 hours away from food, supplements, and medications. This is not optional. Bentonite adsorbs indiscriminately: it doesn’t distinguish between lead and your thyroid medication. It will bind both. If you take prescription drugs, talk to a doctor before using any enterosorbent clay.
Two to four weeks, then stop for a while. Continuous daily use has no clinical safety data behind it. The wellness community runs longer, sometimes indefinitely. I’d want more human data before committing to that. Constipation is also the most common complaint in community forums about extended use. That alone should give you pause.

Bentonite targets heavy metals. For fluoride specifically, tamarind extract has stronger evidence. For calcium deposits, K2+D3 addresses the underlying deposition mechanism more directly. If you want to cover all the bases on pineal health, bentonite is one tool, not the whole protocol. (See the full comparison below.)
This distinction matters more than most people realize. Industrial bentonite goes into drilling fluids, cat litter, and construction. Contaminant loads differ. Look for “food-grade” or “USP-grade” on the label, with third-party testing for heavy metals. Yes, the clay that removes heavy metals needs to be tested for heavy metals. That’s the world we live in.
| Method | What It Targets | Evidence Quality | Notable Risk |
|---|---|---|---|
| Bentonite clay | Heavy metals (GI tract) | Moderate (indirect) | Aluminum content |
| Tamarind | Fluoride (systemic) | Moderate–Strong | Minimal |
| Boron | Fluoride + calcium | Moderate | Dose-dependent |
| K2 + D3 | Calcium redirection | Strong | Minimal at standard doses |
For a full breakdown of decalcification approaches and how they stack, see the complete decalcification protocol guide, the best-studied supplements for pineal support, and how to decalcify your pineal gland naturally.
Bentonite fills a specific niche, heavy metal interception, that the other methods don’t address. It’s not competing with K2+D3. They’re solving different parts of the same problem.
Bentonite clay does not decalcify your pineal gland. That claim outpaces the evidence by about three logical steps.
What it does, if food-grade, used short-term, and timed correctly away from medications, is reduce heavy metal absorption in the GI tract. Over time, less lead and aluminum entering circulation means less available for neural deposition. That’s a real mechanism. It’s just slower, more indirect, and less dramatic than “open your third eye.”
It’s a prevention tool, not a reversal. Part of a protocol, not a solution.
If you want to start somewhere concrete: fix your sleep first. Melatonin production, the primary measurable output of the pineal gland, is almost entirely dependent on sleep hygiene. No clay protocol outperforms eight hours of dark, consistent sleep. That’s not a romantic answer. It’s just what the data says.

Marcus Hale is an independent researcher and former clinical neuroscientist. The content on PinealCode.com is for informational purposes only and does not constitute medical advice.

61.65% of adults have pineal calcification — and the internet promises a 30-day fix. Actual research timeline: 3–6 months. Week-by-week...
Can pineal gland calcification be reversed? Yes — with important caveats. See what the research shows, what works, and realistic timelines.
Only 5 ingredients pass the evidence check for pineal detox. Tamarind reduced fluoride 37% in a human trial. Here's the real protocol — and...

Marcus Hale
Independent Researcher · Former Clinical Neuroscientist
I spent 12 years in clinical neurology before the questions got more interesting than the answers. PinealCode is where I document what I find at the intersection of brain science and consciousness.