
Best Supplements for Pineal Gland Health: Ranked 1–10 by Evidence Score
10 pineal gland supplements ranked by evidence score — not marketing claims. Magnesium scored 8.4/10. Most popular products ranked lower...
NMN has become the longevity supplement everyone is talking about. NAD+ precursor. Anti-aging molecule. David Sinclair’s favorite topic at dinner parties.
What almost nobody is talking about is the nmn pineal gland connection — a mechanistic chain that links NMN’s core mechanism directly to how your brain manufactures melatonin as you age. It’s not a stretch. It’s biochemistry. But it’s also not proven in the way most supplement marketing implies, and I’m not going to pretend otherwise.
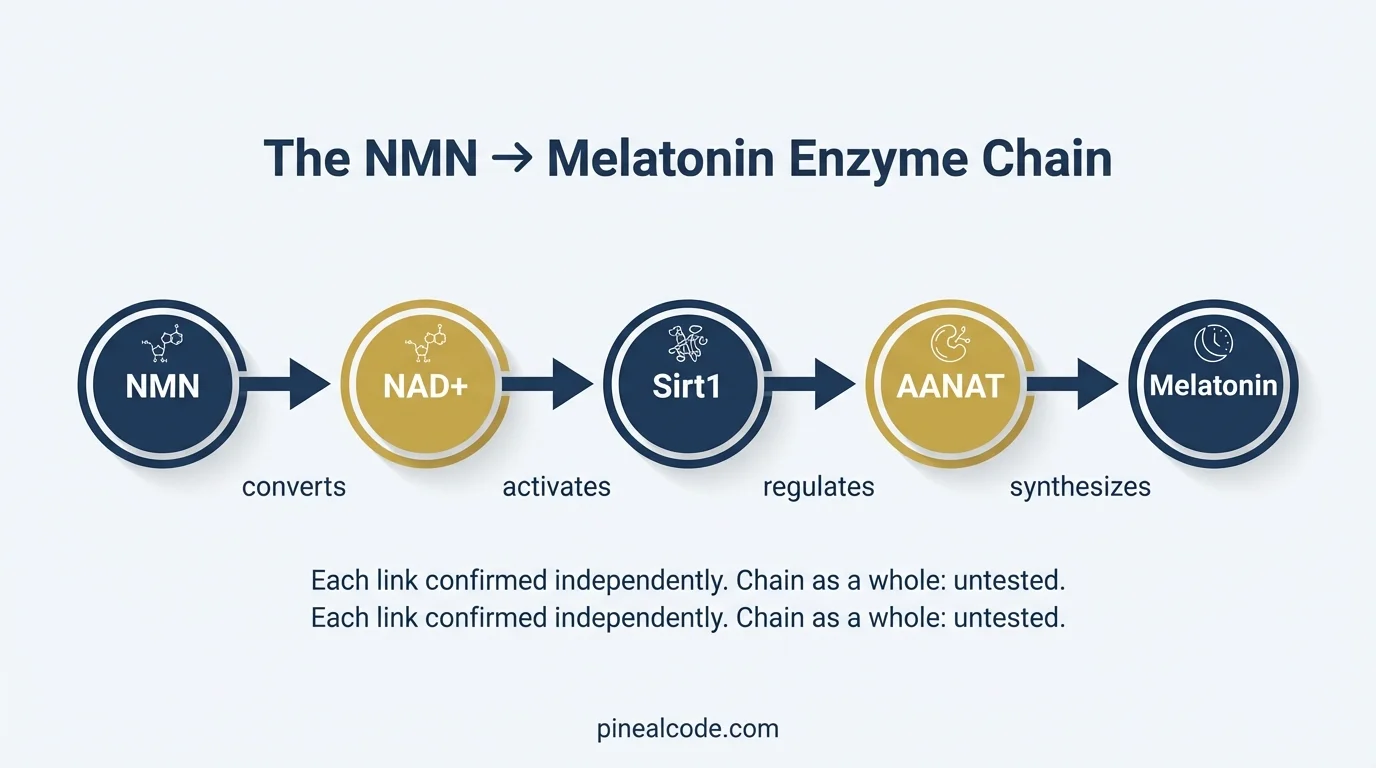
Here’s what the chain actually looks like: NMN raises NAD+. NAD+ activates Sirt1. Sirt1 influences the enzyme that controls melatonin synthesis in your pineal gland. That enzyme, AANAT, requires NAD-dependent cofactors to function. When NAD+ drops with age, the whole chain slows down.
No study has tested NMN directly on pineal tissue. Not in mice. Not in humans. That gap matters, and it’ll come up repeatedly in this article.
If you want to understand which supplements have the clearest decalcification evidence, the best supplements to decalcify the pineal gland breakdown is worth reading first — it gives you the full context for where NMN fits in the stack.
The connection is metabolic, not mystical. NAD+ (nicotinamide adenine dinucleotide) is a coenzyme your cells use in hundreds of reactions, including energy production, DNA repair, and the activation of sirtuins, proteins that regulate cellular aging.
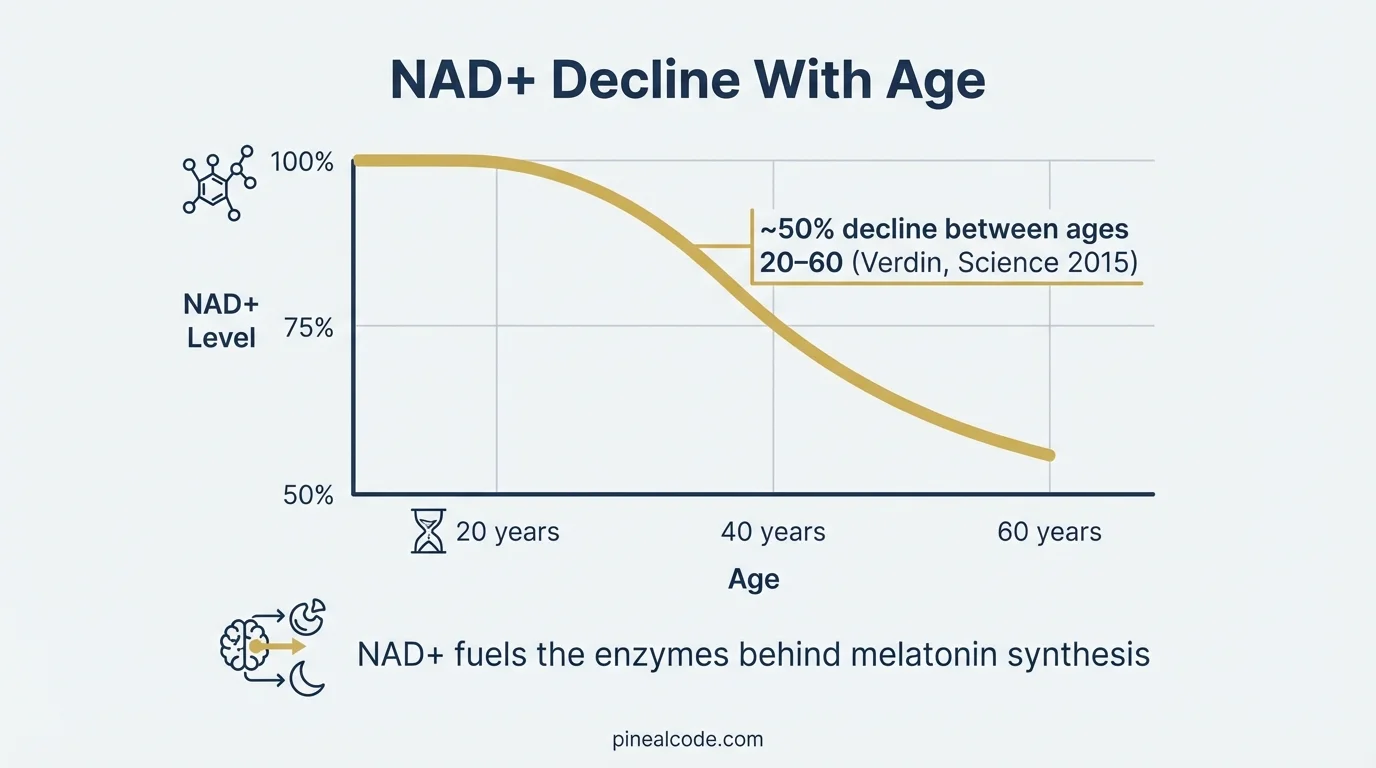
The problem: NAD+ doesn’t stay stable over your lifetime. A 2015 review by Eric Verdin in Science documented that NAD+ declines approximately 50% between ages 20 and 60 across multiple tissues in mammals, including brain tissue. The decline is accelerated by chronic inflammation and oxidative stress, both of which accumulate with age.
That’s where NMN enters. Oral NMN at 250 mg/day was shown in a 2021 Science study by Yoshino et al. to measurably raise plasma NAD+ in postmenopausal women, confirming the precursor reaches systemic circulation and converts. An earlier 2016 study by Mills et al. in Cell Metabolism showed NMN administration in aging mice restored NAD+ levels in liver, skeletal muscle, and brain tissue, reversing several age-associated physiological markers.
The pineal gland is neural tissue. It’s metabolically active. It runs on the same biochemistry.
Which means if NAD+ is declining system-wide, the pineal gland is not exempt.
AANAT (arylalkylamine N-acetyltransferase) is the rate-limiting enzyme in melatonin synthesis. Every night, your pineal gland converts serotonin into melatonin. AANAT controls the speed of that conversion.
The enzyme doesn’t work in isolation. It operates within a protein complex regulated by cAMP-mediated phosphorylation, and its activity depends on NAD-dependent cofactors — specifically acetyl-CoA, which sits upstream in a pathway that requires functional NAD+ metabolism. David Klein at NIH, who spent decades characterizing AANAT’s role, described it as the primary amplitude regulator of the circadian melatonin rhythm.
I realize that sounds like stretching a mechanism to fit a hypothesis. Maybe. But cofactor dependency isn’t speculation — it’s basic enzymology. If NAD+ drops systemically with age, and the data says it does, the enzymatic machinery behind melatonin synthesis is operating under resource constraints.
Not directly. That distinction matters more than most supplement copy wants you to think. NMN does not contain melatonin, does not mimic melatonin, and does not act on melatonin receptors. No human trial has measured melatonin as a primary outcome in NMN supplementation studies. Not Yoshino 2021. Not Huang 2022. The question remains formally unanswered.
What NMN may do is restore the substrate the melatonin-producing enzyme needs to function. The factory analogy holds: NMN restores the fuel supply. Whether the factory then produces more melatonin depends on the overall condition of the factory — enzyme expression, circadian signaling, and the structural integrity of the gland itself.
A 2019 study found that NMN and melatonin together produced greater neuroprotection against aging-induced memory impairment than either compound alone in an animal model. Interesting. It suggests the two may be synergistic at the cellular level, NAD+ amplifying the downstream effects of melatonin rather than replacing it. But it says nothing about whether NMN raises melatonin production.
That’s a smaller claim. It’s also the honest one.
The chain, spelled out:
Each link confirmed independently. None tested as a complete sequence.
I want to be precise about that last point: because the chain is worth taking seriously, and incomplete enough that you shouldn’t rebuild your sleep stack around it.
No study has tested this directly. That’s the honest answer in one sentence. No imaging trial, no biopsy analysis, no CT scan before and after NMN — nothing confirming that NMN reverses or slows calcification in the pineal gland specifically.
What does exist is a mechanistic argument worth unpacking carefully.
Calcification in the pineal (the gland calcifies in roughly 40% of adults by age 30, and in 70–80% of adults over 60 in Western populations) is driven partly by oxidative stress and chronic low-grade inflammation. A 2025 study on NMN in a D-galactose-induced aging mouse model found that NMN at 250–500 mg/kg increased SOD and CAT antioxidant enzyme activity, suppressed TNFα and IL-6, and reduced neuronal apoptosis via activation of the Sirt1/AMPK/PGC-1α axis.
Here’s what that means practically: TNFα, one of the inflammatory markers NMN suppressed in that study, directly inhibits AANAT expression in pinealocytes. So NMN, via Sirt1, may be working against the exact inflammatory signal that suppresses melatonin synthesis in an aging, inflamed pineal gland.
That chain: NMN → NAD+ → Sirt1 → reduced oxidative stress and neuroinflammation → potential inhibition of calcification progression → preserved AANAT function.
Mechanistically plausible. Not clinically proven. I keep writing that disclaimer and wondering if I’m being too conservative — the mechanism really is unusually coherent for a supplement chain. But I’ve also said that about compounds that never made it past mouse models. Same honest caveat that applies to NAC and the pineal gland: the antioxidant angle is real, the direct pineal evidence is not.
If you want a broader review of what interventions have actual decalcification data, how to reverse pineal gland calcification goes deeper into the literature.
Sirt1 is not a supplement. It’s an enzyme your cells produce — a deacetylase that coordinates the cellular response to metabolic stress. When NAD+ is sufficient, Sirt1 stays active. When NAD+ drops, Sirt1 activity declines, and the cell’s antioxidant defenses degrade.
In neural tissue, Sirt1 governs PGC-1α, which controls mitochondrial biogenesis and oxidative stress response. In the context of the pineal, this matters because pinealocytes are metabolically demanding — running a high-output synthesis cycle every night. Chronic oxidative damage in that environment accelerates structural deterioration, including calcification.
NMN → NAD+ → Sirt1 is a confirmed chain in neural tissue models. The extension to pineal calcification is the inferential step. Biologically coherent, experimentally untested.
The short answer: it doesn’t need to. The pineal gland sits outside the classic blood-brain barrier — it’s one of the circumventricular organs, structures with fenestrated capillaries and direct vascular access. Compounds in systemic circulation reach the pineal without crossing the BBB at all.
So for the pineal specifically, the question as usually framed is the wrong question.
For broader brain tissue, the evidence is cautiously positive. A 2019 study by Grozio et al. in Nature Metabolism identified the Slc12a8 transporter as a direct uptake route for NMN in intestinal and brain tissue in mammals, suggesting NMN enters cells as an intact molecule rather than only after conversion to NR. The finding remains partially contested — the debate about pre-absorption conversion to NR is ongoing — but the direction of evidence supports CNS tissue uptake.
Sublingual NMN formulations add another layer. Work from Okabe et al. (2022) showed sublingual delivery produced a faster plasma NAD+ peak compared to standard oral administration, bypassing first-pass hepatic metabolism. Faster systemic elevation means faster availability to tissues, including the pineal’s direct vascular supply.
The access pathway looks favorable. What happens after access is the open question.
NMN is one of several candidates with mechanistic rationale. None of them has confirmed anti-calcification effects in human imaging trials — and I’d be doing you a disservice by implying otherwise.
Here’s how the main players compare:
| Supplement | Mechanism | Evidence Level | Pineal Angle |
|---|---|---|---|
| NMN | NAD+→Sirt1→AANAT | Mechanistic (indirect) | Melatonin synthesis, antioxidant |
| NAC | GSH replenishment | Mechanistic (indirect) | Antioxidant, same honest caveat |
| Boron | Fluoride displacement | Observational | Decalcification direct |
NAC and NMN share the same fundamental angle: both target oxidative stress via different upstream pathways. NAC and the pineal gland compared is worth reading if you’re deciding between them or considering a stack.
Boron’s evidence is different in character — more direct, though still observational. The full supplement protocol for decalcification puts all three in context with the complete evidence hierarchy.
Yes, there’s a supplement for every mechanism. There’s always a supplement for that. The question is whether the mechanism is real and whether the dose in the capsule matches the dose in the study.

Human trials have used 250–500 mg/day. Yoshino et al. 2021 used 250 mg/day for 10 weeks in postmenopausal women and confirmed plasma NAD+ elevation. The Huang et al. 2022 multicentre RCT worked in the 250–300 mg range. Those are the doses with actual human safety and efficacy data behind them.
Sublingual delivery shows faster uptake kinetics — relevant if your hypothesis is timing the NAD+ elevation to support the nighttime AANAT synthesis window. That timing hypothesis (NMN taken in the evening, synergistic with the circadian melatonin cycle) is biologically plausible but untested. There are no pharmacokinetic studies comparing morning vs. evening NMN on melatonin output.
Practically: start at 250 mg, standard oral or sublingual, and treat it as a long-term metabolic investment rather than an immediate sensory intervention. If you’re expecting to feel anything acutely, you’ll be disappointed. The benefit, if it exists for the pineal, is enzymatic and preventive — not something you’ll notice on night two.
Consult a healthcare provider before starting any NAD+ precursor supplement, particularly if you’re on medications affecting sirtuin pathways or have a history of hormone-sensitive conditions.

The NMN–pineal chain is the most mechanistically coherent argument I’ve come across in the NAD+ space that actually connects to sleep quality and melatonin synthesis. NAD+ decline is real. AANAT’s dependency on NAD-related cofactors is established biochemistry. Sirt1’s neuroinflammatory role has solid animal data behind it.
What doesn’t exist yet: a single study testing NMN in pineal tissue, measuring AANAT activity, or showing melatonin output changes in a clinical population. The chain is built from solid links. The chain as a whole is untested.
There’s a version of this story that ends with NMN being the longevity compound that actually worked across the board — the NAD+ version of what Linus Pauling wanted Vitamin C to be, but with better trial data. There’s another version where it joins the long list of mechanistically compelling compounds that never replicated in humans at real-world doses. I genuinely don’t know which story we’re in yet.
If you’re going to take NMN, take it for the systemic NAD+ evidence, which is solid. The pineal angle is a reasonable hypothesis for why sleep-related benefits get reported anecdotally. Don’t take it expecting to see calcification reverse on a follow-up scan.
Start with sleep hygiene and consistent darkness exposure. Those directly drive the AANAT activation cycle that NMN is hypothetically supporting. Build your supplementation around that foundation, not the other way around.
Marcus Hale is an independent researcher and former clinical neuroscientist. The content on PinealCode.com is for informational purposes only and does not constitute medical advice.

10 pineal gland supplements ranked by evidence score — not marketing claims. Magnesium scored 8.4/10. Most popular products ranked lower...
One peer-reviewed study found 9,000 ppm of fluoride in human pineal glands — 18,000x more than in muscle. Here's what that data means, what...
61.65% of adults have pineal calcification — and the internet promises a 30-day fix. Actual research timeline: 3–6 months. Week-by-week...

Marcus Hale
Independent Researcher · Former Clinical Neuroscientist
I spent 12 years in clinical neurology before the questions got more interesting than the answers. PinealCode is where I document what I find at the intersection of brain science and consciousness.